The CE Marking process
The CE Marking Process has six simple, but necessary steps to complete to enable you to CE Mark your product.
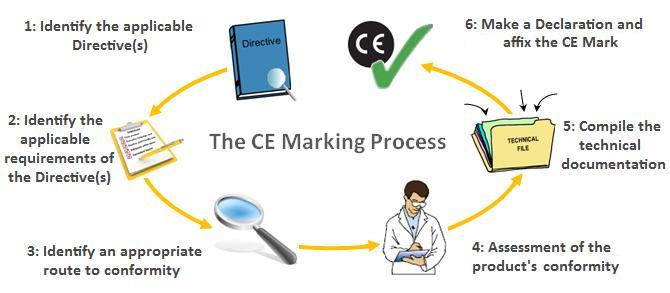
Step 1: Identify the applicable Directive(s)
The first step is to identify whether your product can be CE marked or not. Not all products are required to be CE Marked, only the products that fall within the scope of at least one of the CE Marking Directives. There are more than 20 product Directives & Regulations covering a range of products. Such products includes (but not limited to) electrical equipment, machines, medical devices, toys, pressure equipment, PPE, wireless devices and construction products.
Identifying which Legislation may be applicable involves a simple exercise of reading the scope of each Directive/Regulation. You then need to apply the scope to your product to establish which ones applies. (An example of the scope of the Low Voltage Directive below). Don’t forget that there may be more than one Directive/Regulation that applies to your product! Remember, if your product does not fall within the scope of any of CE marking Directives, then your product does not need to be CE marked, but is likely to fall within the scope of other product Directive(s).
Article 1 states the Directive covers “any equipment designed for use with a voltage rating of between 50 and 1000 V for A.C. and between 75 and 1500 V for D.C, other than the equipment and phenomena listed in Annex II.”
Step 2: Identify the applicable requirements of the Directive(s)

Each Directive has slightly different methods of demonstrating conformity. This usually depends on the classification of the product and its intended use. Every Directive has a number of ‘essential requirements’ which the product has to meet.
The best way to demonstrate that these essential requirements have been met is by meeting the requirements of applicable ‘harmonised European Norms’ (Standard) known as hENs. Standards may offer a presumption of conformity to the essential requirements of the applicable Legislation. However do not forget that the use of standards usually remains voluntary. hENs can be identified by searching the ‘official journal’ on the European Commission’s website.
Step 3: Identify an appropriate route to conformity
The CE Marking process is always a self-declaration process however you may need to involve a third party. This is set out in the ‘system of attestation’ and will vary between Directive. Some products (such as invasive medical devices or fire alarm systems) may, to some extent, have a mandatory requirement for some involvement of an authorised third party.
There are various attestation routes which include:
- An assessment of the product by the manufacturer.
- An assessment of the product by the manufacturer, with additional requirement for mandatory factory production control audits to be carried out by a third party.
- An assessment by a third party (e.g. EC type test), with the requirement for mandatory factory production control audits to be carried out by a third party.
Step 4: Assessment of the product’s conformity
 When all of the requirements have been established, you need evidence that the product meets the essential requirements of the Directive(s).
When all of the requirements have been established, you need evidence that the product meets the essential requirements of the Directive(s).
This usually involves some assessment and/or testing. It will often involve ensuring that the requirements of the applicable harmonised standard(s), which were identified in step 2, have been met.
Step 5: Compile the technical documentation
Technical documentation relating to the product or range of products needs to be compiled. This information should cover every aspect relating to conformity and is likely to include details of the design, development and manufacture of the product. Technical documentation may also be known as the Technical File or Technical Construction File.
- Technical description.
- Drawings, circuit diagrams and photos.
- Bill of materials.
- Specification and, where applicable, Declarations of Conformity for the critical components and materials used.
- Details of any design calculations.
- Test reports and/or assessments.
- Instructions.
- Copy of the Declaration.
Technical documentation can be made available in any format (i.e. paper or electronic) and must be held for a period of up to 10 years after the manufacture of the last unit, and in most cases reside in the European Economic Area (EEA).
Step 6: Make a Declaration and affix the CE Mark
When the manufacturer, importer or authorised representative is satisfied that their product conforms to the applicable CE Marking Directives, they must complete a Declaration. Under most Directives it is known as the EU Declaration of Conformity but other terms exist. Such as Declaration of Incorporation for partly completed machinery and Declaration of Performance for construction products.
The requirements for the Declaration vary slightly, but will at least include:
- Name and address of the manufacturer.
- Details of the product (model, description and the serial number where applicable).
- List of CE Marking Directives and standards that have been applied.
- A statement declaring that the product complies with all of the relevant requirements.
- Signature, name and position of the responsible person.
- The date that the Declaration was signed.
- Details of the authorised representative within the EEA (where applicable).
- Additional Directive/standard specific requirements.
In all cases, except for the PPE Directive, all of the Directives can be declared on one Declaration.
Once a Declaration has been completed, the final step is to affix the CE mark to the product. When this has been done and all of the other CE Marking requirements have been met, the product can then be legally placed on the EU market.
If you need help with any part of the CE marking process or any aspect of product compliance, then please call us on 01564 792349.










